The stem was so long that it couldn’t support itself…

Some flowers up close!

And this is the rosette what did it:

Have a good weekend!
Just another plant science blog

The stem was so long that it couldn’t support itself…

Some flowers up close!

And this is the rosette what did it:

Have a good weekend!

And now, for something a bit different.
We’ve got this jasmine bush growing indoors under a growlight. Recently, some of its leaves started to turn yellow and drop off.

Figure 1. A sad jasmine bush. And it’s covered in mysterious sachets. Hm…
Oh no!
At first, I thought it might be a watering or nutritional issue. However, I discovered another problem upon closer inspection. Suspecting that something might be feeding on the plant’s precious bodily fluids, I broke off a piece of leaf to look at on the microscope and it was teeming… with mites! Here’s one on the move:
Video 1. Mite on a jasmine leaf.
I’m not an arachnologist, but these appear to be two-spotted spider mites. They are common pests which can cause damage to a wide variety of plant species [4]. I watched the mites for a few minutes. During this time, I observed them moving from place to place, occasionally stopping to feed on the leaf.
I wanted to get a close-up video of a mite feeding, but there was a slight problem. The light source on my microscope is not sufficient for this purpose. This is for two reasons:
The solution is simple: use a brighter light source. Fortunately, I have an LED bulb on hand which is much brighter than my usual light source. So, into the microscope it went!

Figure 2. Top: Opening the bottom of the microscope to replace the light source. Bottom left: Original bulb before replacement. Bottom right: New LED light source. Much better!
I don’t normally use the LED light because it can be too bright (hurts my eyes) and has a harsh blue-ish color. However, it was quite useful in this case.
Right, then… behold! A spider mite feeding on a jasmine leaf!
Video 2. Spider mite feeding on a jasmine leaf.
If you look closely, you can see a bunch of… stuff inside the mite moving from side to side. What is it doing? According to this source [3], this material is located inside of the caudal caeca. The caudal caeca are two sac-like extensions of the gut that run along the sides of the mite’s body. They connect to each other in the center of the mite’s body, just behind the head. Contraction of muscles around the gut causes material to move rhythmically from one caecum to the other [3].
The little spots we can see moving back and forth are probably digestive cells. These specialized cells break off from the lining of the caeca. Then, they begin to ingest material from the surrounding gut contents as they float freely about. They are believed to accumulate unwanted digestive waste products. Consistent with this idea, they are eventually excreted as faecal material [3]. Pretty neat! In case my description was not colorful enough, Figure 1 from the source paper [3] is extremely helpful.
Fighting mites with mites
Given that these spider mites are damaging my jasmine plant, how can I stop them?? This plant is indoors (and it is winter, so it can’t go outside). Therefore, physical measures (spraying with a hose to remove the mites) and pesticides aren’t super practical. Removing the more heavily infested branches seems like a good place to start. A biological pest control solution might also also help. In this case, I have decided to fight the spider mites with yet more mites. Sure, that seems reasonable…
To be specific, I’ve employed the help of a predatory mite, Amblyseius andersoni, which actually eats spider mites. How convenient! They come in little sachets which resemble tea bags. You hang the sachets on the infested plant, and the Amblyseius mites crawl out of the little hole in the sachet and begin to feed on the spider mites. They might not finish off the spider mites completely, but I hope they will help me to keep them under control until I can get the plant outside (where I can use other control techniques if necessary).
As it happens, I received way more sachets than I ordered. So, rather than let them go to waste, I absolutely covered the jasmine bush with them (See Figure 1). This is probably overkill, but we’ll see.

Figure 3. A sachet containing Amblyseius andersoni mites.
There are a number of different predatory mite species which are commercially available to help deal with spider mite infestations. I don’t know much about mites, but I chose A. andersoni (as opposed to, say, Persimilis mites) because it is supposed to be able to tolerate a wider range of temperature and humidity conditions [2][5]. This is necessary because our living space is fairly cool (~66˚ F) and dry (~40% RH), and predatory mites usually prefer higher temperature and humidity. To help matters, I’ve placed a humidifier next to the jasmine bush to make the mites more comfortable.
After receiving my A. andersoni in the mail, I decided to cut one of the sachets open to see if the mites were viable. I was pleased to see many live mites at several different life cycle stages. Mites are arachnids, meaning that they have 8 legs. However, the larval stage of A. andersoni only has 6 legs [1]. Here is one that I saw:
Video 3. A. andersoni larva, with 6 legs.
As the mites grow and molt, they develop their final pair of legs. This mite is at a later stage of the development process and has all 8 legs. They’re darn quick, too, and really tricky to film… I got lucky with this one!
Video 4. A more mature A. andersoni mite, now with all 8 legs. New-and-improved!
Here’s hoping our mite friends do the trick!
Works Cited
[1] Amblyseius andersoni. (2025). Koppert US.Retrieved February 15, 2026, from: https://www.koppertus.com/crop-protection/biological-pest-control/predatory-mites/amblyseius-andersoni/?utm_term=redirect
[2] Amblyseius andersoni – Generalist, Tolerates High Heats. (2026) NaturesGoodGuys. Retrieved February 15, 2026, from: https://www.naturesgoodguys.com/products/amblyseius-andersoni?variant=42422847111359
[3] Bensoussan, N., Zhurov, V., Yamakawa, S., O’Neil, C.H., Suzuki, T., Grbić, M., & Grbić, V. (2018). The Digestive System of the Two-Spotted Spider Mite, Tetranychus urticae Koch, in the Context of the Mite-Plant Interaction. Frontiers in Plant Science 9, 1206.
[4] Hazzard, R. (2022, July 28). Two-Spotted Spider Mite. UMassAmherst. Retrieved February 15, 2026, from: https://www.umass.edu/agriculture-food-environment/vegetable/fact-sheets/two-spotted-spider-mite
[5] Murray, M. (n.d.). Greenhouse Biocontrol in Utah. Utah State University. Retrieved February 15, 2026, from: https://digitalcommons.usu.edu/cgi/viewcontent.cgi?article=3332&context=extension_curall
CORRIGENDUM
In an earlier iteration of this blog post, Video 4 was the video shown here (which we will shall now call “Video 5”). This is NOT A. andersoni, but is instead an unidentified species of mite – likely included as food for A. andersoni in the sachets. Video 4 now shows A. andersoni. (The long legs are a hint).
Video 5. An unknown mite, which I mistakenly labelled A. andersoni…

Last week I introduced you to the genus Lithops, which have a fairly unusual anatomy. While observing my own lithops plants over the last several weeks, I made a few more miscellaneous observations that I wanted to share with you. I hope you find them diverting!
Otherwise – happy holidays! I look forward to providing more content to you, the reader, in the coming year.
Splitting!
OK, this observation was particularly exciting for me, a first-time lithops owner. I noticed that one of my plants has started splitting! This is the process by which lithops grow. It looks like this:

Figure 1. A splitting lithops plant!
What are we looking at here? As we learned last week, when you look at a lithops plant, you can see two fused succulent leaves. New sets of leaves form deep inside the center of the plant, and are not initially visible (See Figure 2 left). As the new leaves grow, they push the old leaves apart. To conserve water, the new leaves will absorb water from the old leaves; this is a helpful adaptation to living in dry climates [4]! Eventually, the old leaves shrivel and die, leaving just the new pair of leaves behind.
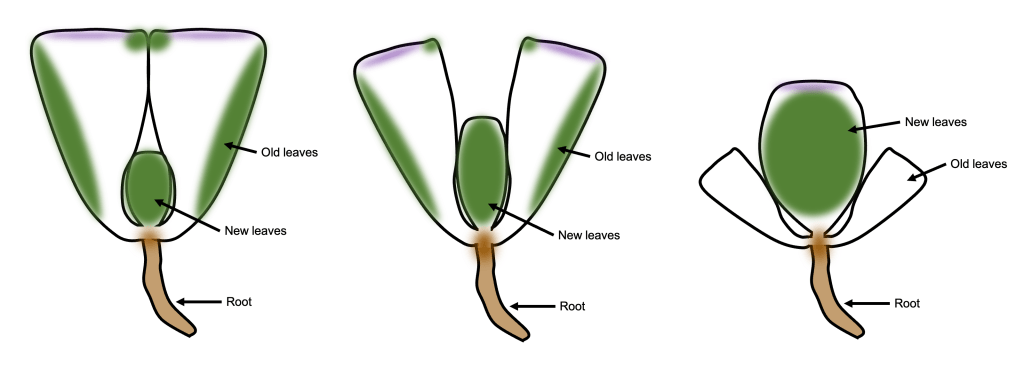
Figure 2. Simplified diagram of the splitting process.
Itchy scratchy…
One of the things I noticed while looking at lithops tissue on the microscope was the presence of lots of tiny crystals. These were particularly abundant when I squished the tissue (which releases cell contents into the water that the tissue is suspended in). These crystals are very difficult to see under normal illumination (Figure 3 left). However, using polarized light microscopy, their presence becomes… crystal clear (Figure 3 right)!
(For the polarized light microscopy, I ordered a cheap polarizing filter sheet online and cut it into two halves. I placed one half beneath my sample, and the other half above it. Then, I rotated the top polarizer so that it was perpendicular to the bottom polarizer. This is super easy to do at home! I won’t say much more here, but there is a really excellent animation explaining the principle behind polarized light microscopy on the relevant Wikipedia article: https://en.wikipedia.org/wiki/Polarized_light_microscopy).

Figure 3. Raphides from damaged lithops tissue. Left: Under normal illumination. Right: Using polarized light microscopy.
From my reading, I am reasonably confident that we are looking at raphides here. Raphides are tiny crystals (typically calcium oxalate) which are common in many species of plants. They are thought to protect plants against predation, as they are sharp and could in theory cause irritation in animal tissues that come in contact with them.
I found this interesting study [2] which demonstrates that raphides inhibit the growth of silkmoth larvae when ingested, particularly in combination with cysteine protease, an enzyme common in plant tissues. Importantly, this study verified that the shape of the raphides, rather than their chemical composition, contributes to their defensive function (since amorphous calcium oxalate particles did not have the same effect) [2]. Fascinating. There is apparently also evidence that raphides in large quantities are toxic to humans. This article about an outbreak of foodborne illness in Chicago associated with raphide consumption made for particularly grim reading [6].
I couldn’t find much information about raphides in lithops specifically, although the thesis of Robert Wallace (Rutgers University, 1988) does note their presence [5]. So, someone has seen this before! Whew.
AAAAAAAAAAAAH
Stomata are pores found on virtually all plants which facilitate gas exchange with the environment. Plants take up oxygen from the atmosphere for respiration and CO2 for photosynthesis. Conversely, oxygen is also released from plant tissues as a byproduct of photosynthesis, and CO2 is released as a byproduct of respiration. Thus, stomata help plants to achieve the correct concentration of oxygen and CO2 in their tissues. Stomata also allow plants to transpire (release water via evaporation), which is essential for thermoregulation and for water transport. I might do a dedicated post (or a few!) about stomata later – they’re really interesting!
Plants control gas exchange by opening and closing their stomata. This opening/closing is facilitated by specialized cells known as guard cells. Two guard cells lie on either side of every stomatal pore. The pore opens when the guard cells inflate, and closes when the guard cells deflate (See Figure 4). Guard cells inflate/deflate due to intake/expulsion of water via osmosis. How this is controlled is beyond the scope of this post, but I may come back to this at a later date.
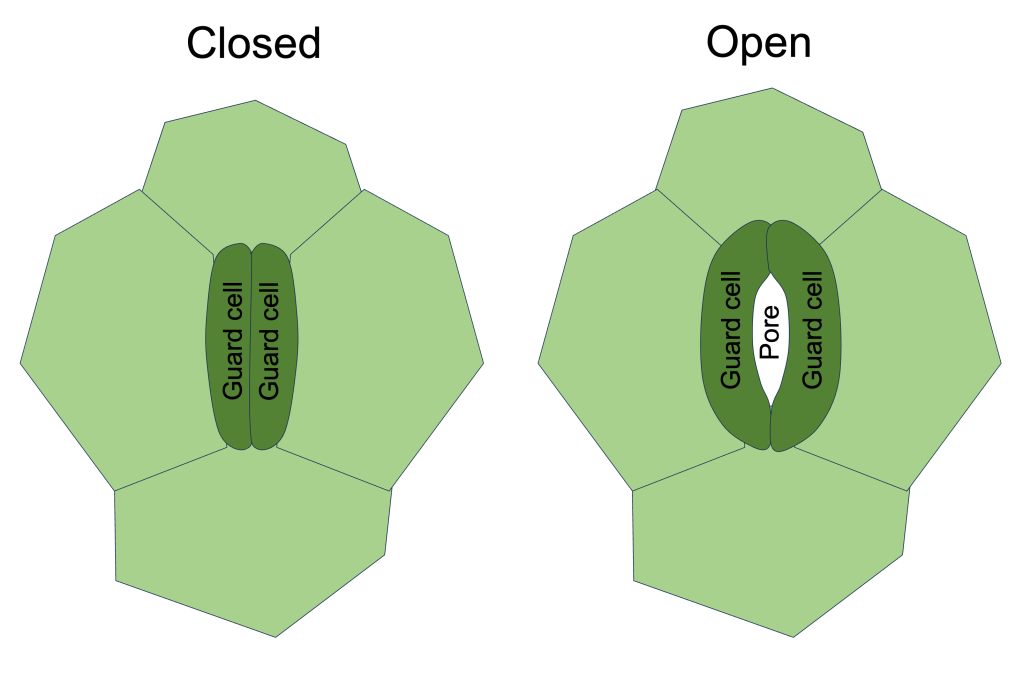
Figure 4. Simplified diagram of a single stoma in the closed state (left) and open state (right).
Why am I talking about stomata now? I am talking about stomata because I came across this interesting blog post [3] a short while ago. It’s a great piece on lithops biology (would recommend!), but one sentence stood out to me: they claim that one species, Lithops dorotheae, has 3 guard cells per stoma instead of the usual 2! This was surprising to me, and raised a few questions:
I decided that it would be fun/worthwhile to verify this myself. To this end, I did two things: First, I tried to find the original source of this information to see what it said, exactly. Second, I acquired some domestically-grown L. dorotheae plants so that I could make my own observations! Note: when purchasing lithops, it is important that you buy domestically-grown plants, for ecological, health, and legal reasons. Always check the source. Anyway…

Figure 5. A small L. dorotheae plant. It looks a bit different to my other lithops (which I’m pretty sure are L. karasmontana). Check out the little red stripes!
OK, so the source of the information about the unusual guard cells in L. dorotheae appears to be that Wallace thesis from 1988 [5]. However, Wallace says that that stomata in L. dorotheae have 3 subsidiary cells… NOT that they have 3 guard cells. Aha! This makes more sense.
Subsidiary cells are specialized epidermal cells which surround the pair of stomatal guard cells. In the case of my L. karasmontana plant, these cells are indistinguishable from other epidermal cells (Figure 6 left). However, in L. dorotheae, I observed the presence of 3 or 4 cells surrounding each stoma, which are more darkly colored than the other epidermal cells (Figure 6 right). I think this is probably what Wallace was referring to. Importantly, the stomata on my L. dorotheae plant have the normal 2 guard cells… (Figure 7).

Figure 6. Epidermis of my L. karasmontana (?) plant (left) and L. dorotheae plant (right) at 100X magnification. The top half of the figure are the original images; In the bottom half, I have circled the stomata that I can see.
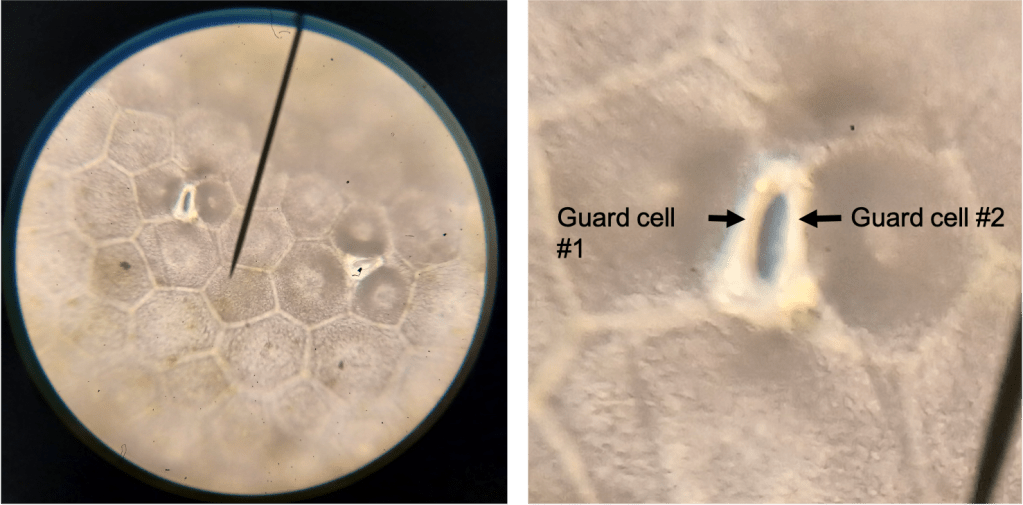
Figure 7. Stomata in L. dorotheae, at 400X magnification. Left is the original image; Right is a cropped version of the same image blown up to show a single stoma clearly. The two guard cells are visible (compare to my drawing in Figure 4).
What do the subsidiary cells actually… do? Unfortunately, this is not an easy question to answer, and probably depends on the species of plant. One thing that Wallace (1988) mentions is that stomata in lithops are sunken below the main surface of the epidermis [5]. The shape of the subsidiary cells may help to achieve this sunken topography. It has been theorized that sunken stomata are an adaptation to prevent excessive water loss in plants growing in dry climates [1]. Having sunken stomata increases the depth of the boundary layer of air above the pore, which may slow the diffusion of water molecules away from the pore [1].
Works cited:
[1] Gray, A., Liu, L., & Facette, M. (2020). Flanking support: How subsidiary cells contribute to stomatal form and function. Frontiers in Plant Science 11. DOI: 10.3389/fpls.2020.00881
[2] Konno, K., Inoue, T.A., & Nakamura, M. (2014). Synergistic Defensive Function of Raphides and Protease through the Needle Effect. PLoS One 9(3), e91341.
[3] Living stones: Growing Lithops. (2018, October 25). Nerd Rambling. Retreived December 21, 2025, at: https://atreyuoz.blogspot.com/2018/10/growing-lithops.html.
[4] Sajeva, M., & Oddo, E. (2007). Water Potential Gradients beween Old and Developing Leaves in Lithops (Aizoaceae). Functional Plant Science and Biotechnology, 1(2), 366-368.
[5] Wallace, R. (1988). Biosystematic Investigation of the Genus Lithops N.E.BR. (Mesembryanthemaceae). PhD dissertation, Rutgers University.
[6] Watson, J.T., Jones, R.C., Siston, A.M., Diaz, P.S., Gerber, S.I., Crow, J.B., & Satzger, R.D. (2005). Outbreak of food-borne illness associated with plant material containing raphides. Clinical toxicology 43(1), 17-21.

You may have noticed that there was a large gap between this post and the last. My apologies. I’m increasingly busy, and so my posts will unfortunately be more sporadic from now on. But fear not, the releases shall continue! They’ll be special occasions… think Christmas, but not as good.
Anyway, I recently acquired these lovely lithops plants at a local garden center (Figure 1 left). The name “lithops” is derived from the Greek word for “stone”, because they look like kind of like pebbles when viewed from above. They’re definitely weird looking. I rather like them, but some of my relations commented that they look like toes. Now I can’t unsee that. **shudders**.
Lithops is a genus of succulents native to southern Africa. Because they are popular houseplants, you should be able to find tons of information about their care and growth habits online. There are also already a few excellent blogs about lithops biology (easily found via google search). Therefore, in order make this blog post a bit more unique, I will be using my light microscope to look at some of their more interesting features up-close! So, read on if you want to see lithops in more detail than you would ever reasonably need to.
An aside: I’ve never seen lithops in the wild, but I noticed while looking through some old pictures that I’d inadvertently photographed one of their relatives in the Canary Islands years ago. Both lithops and the ice plant (Figure 1 right) are members of the family Aizoaceae. And they’re both unusual in their own right!

Figure 1. Left: An unidentified lithops (possibly L. karasmontana?) plant currently inhabiting our hallway. Right: an ice plant (Mesembryanthem sp.) photographed on the Canary Islands. They don’t look very much alike, do they? Try looking for images of their flowers… that might clear things up for you.
Ok, seriously, what am I looking at here?
Looking at Figure 1, it’s probably obvious to you that lithops have a fairly unusual anatomy. They barely look like plants at all! So, when we look at a lithops, what are we really seeing?
Basically, the part of the plant that we can see is made up of two succulent leaves that are fused together at the base. The other parts of the plant, such as new pairs of leaves and the meristematic tissue that generates them, are hidden inside (see Figure 2 below, or source [6] for a photograph of a dissected lithops).

Figure 2. Cutaway of a lithops plant to show its internal structure.
The leaves of the lithops plant have an unusual structure. The green photosynthetic tissue is not visible from the outside; Rather, it lines the inside of the sides of the leaf. The skyward-facing part of the leaf, also known as the “window”, is relatively transparent, allowing sunlight to enter and reach the photosynthetic tissue. Most of the volume of the leaf is taken up by transparent parenchyma tissue, which stores water and allows light penetration.
Despite its odd anatomy, when we examine lithops at the cellular level, we can see that it’s really not so different from other plants after all. For example, if we look at the epidermis of the leaf, we can see that it is made up of tightly interlocking epidermal cells punctuated by pores known as stomata (Figure 3). We’ll talk more about those in our next blog post (stay tuned!). But for now, just know that these structures are found in virtually all land plants and are required for gas exchange between the leaf and its environment (O2 and CO2 for respiration and photosynthesis, respectively).

Figure 3. Closeup of the lithops epidermis (400X), showing epidermal cells and an individual stoma. Stomata are themselves made up of two guard cells, which surround a central pore.
If we cut into a leaf and examine some of the green tissue, we can see that there is a layer of photosynthetic cells which are by traversed by a network of vascular tissue (Figure 4). Taking a closer look at the photosynthetic cells, we can see that there are abundant chloroplasts, which give them their green color (Figure 4 bottom left). If we look more closely at the vascular tissue, we can see intricate reticulations which are characteristic of xylem, which transports water (Figure 4 bottom right). This is all pretty textbook plant stuff, nothing unusual to see here.

Figure 4. Closeup of lithops green tissue. Top right: A zoomed-out image of a section of green tissue. Bottom left: 400X image of photosynthetic cells, showing chloroplasts. Right: 400X image of vascular tissue.
Finally, if we take a transverse section of a leaf, we can observe the transition from the green photosynthetic tissue on the side to the clear tissue of the window on the top (Figure 5). The lack of pigmentation in the cells directly below the window allows sunlight to enter the leaf. However, I did notice a slight purple tinge in this region – this is possibly due to the presence of anthocyanins, which may help protect delicate structures within the leaf from harmful radiation [2]. In Figure 5, we can also observe the water-storing parenchyma cells. These cells lack pigment, and are super-ginormous (technical term).
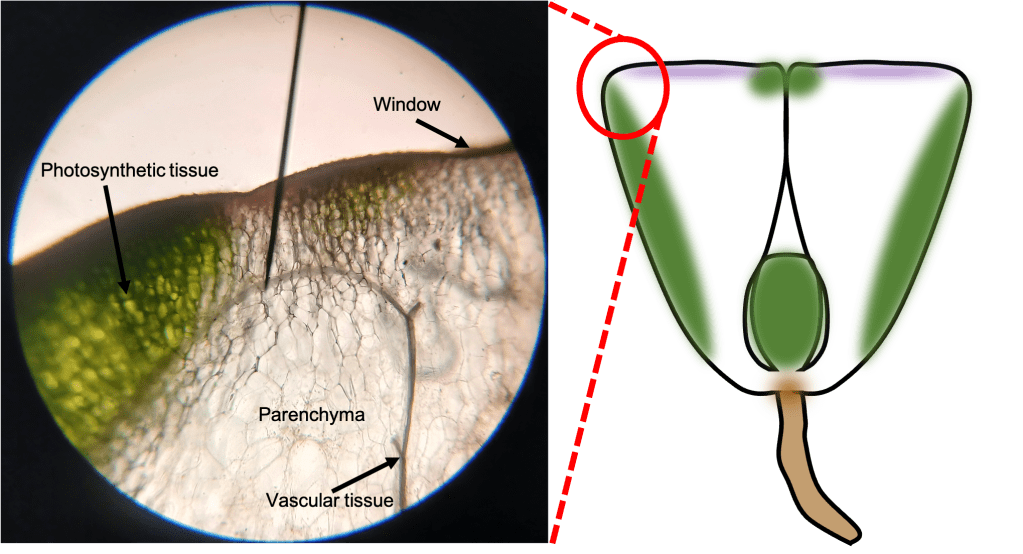
Figure 5. Transverse section of a lithops leaf, showing several different parts: photosynthetic tissue, window, and parenchyma tissue.
“How many are there?! 36, counted them myself…”
The classification of species within the Lithops genus is a bit of a complicated affair. As of 2011, there were actually 37 recognized species of lithops [3], though this number is not set in stone. One problem is that different species of lithops are difficult to tell apart by eye. This is partly due to considerable variation in morphology even within individual species. As such, the grouping of lithops populations into species is subject to potential revision. For example, a genetic study in 2019 determined that L. amicorum was not its own distinct species, but was actually a sub-population of L. karasmontana [5]. (So perhaps there are 36 species now, eh?)
Genome sequencing would surely enable more robust phylogenetic analysis within the Lithops genus. Unfortunately, I could not find any publicly available Lithops genomes by searching the NIH genomes database (https://www.ncbi.nlm.nih.gov/home/genomes/). There appear to be two Lithops genome sequencing projects currently in the works: Lithops lesliei (Iridian Genomes) and Lithops karasmontana (Kew Gardens), but there isn’t much available information from either of these projects yet (although Kew does have some annotated gene sequences for L. karasmontana available on the tree of life: https://treeoflife.kew.org/specimen-viewer).
Of course, genome sequencing is not a trivial exercise; Furthermore, to look at relationships between species, you would need to do it many times over. If only there were a more efficient way to assess the genetic differences between distinct populations of lithops!
The two studies that I cited above [3][5] do just that, using something called AFLP markers (Amplified Fragment Length Polymorphisms). But what the heck are AFLP markers? Fortunately, the name “AFLP” is fairly descriptive. AFLP’s allow you to differentiate between populations by assessing the Lengths of Amplified Fragments. Er…
When we say “Fragments”, we are referring to pieces of DNA. You can generate DNA fragments by digesting genomic DNA with restriction enzymes – which cut the DNA in a sequence-specific manner. This produces many fragments of variable lengths. Genomic DNA from different species varies in the location and number of restriction sites, and so will generate fragments of different sizes.
When we say “Amplified”, we mean that the DNA fragments have been amplified using a technique called PCR. Basically, PCR allows you to take tiny amount of DNA and copy it over and over again until it can be easily detected. Amplified DNA fragments are then sorted by size using a technique called electrophoresis, and are subsequently detected using specialized photography. Advanced statistical techniques are then used to assess the similarity between species on the basis of the sizes of many DNA fragments simultaneously.
A more detailed explanation of AFLP markers is beyond the scope of this blog, but I’ve added a grossly over-simplified diagram below to make the concept easier to visualize. I’ll also direct you to this short article, which explains AFLP’s better than I ever could: [1]. But the take-home message is this: AFLP markers have enabled researchers to explore the genetic relationships between Lithops species without the need for genomic sequencing. And that’s a wonderful thing!
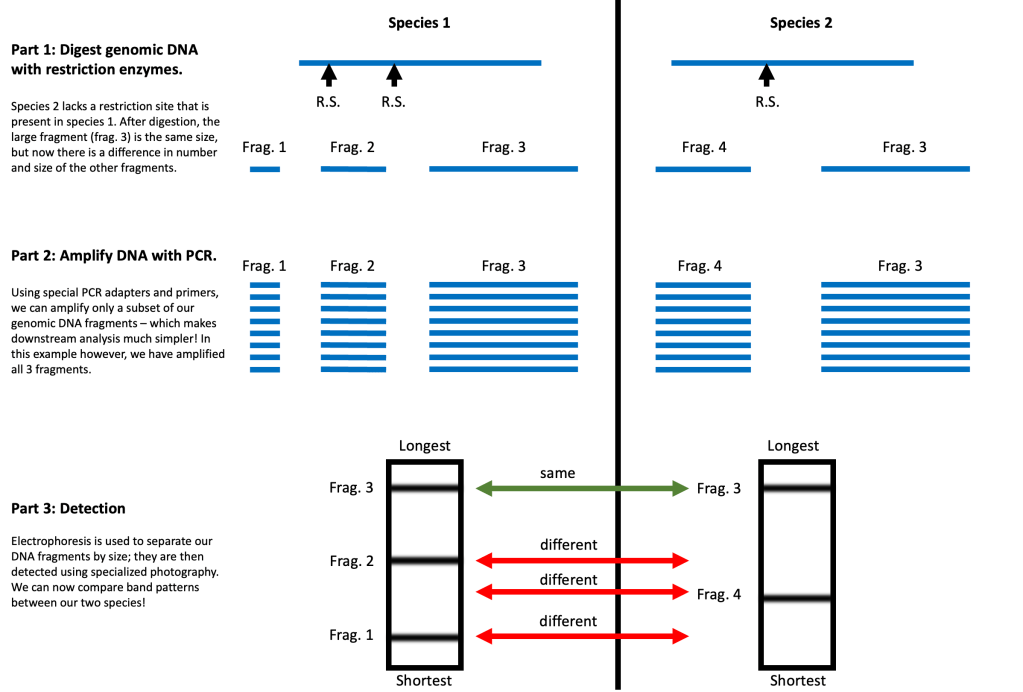
Figure 6. Simplified overview of AFLP marker analysis to assess the genetic similarity between two species.
Gene of the week/month/or something:
Let’s conclude this first exciting installment of our “Lithops” series with a Gene of the Week! As I pointed out earlier, there are not annotated genomes available for Lithops, and the functions of individual genes in Lithops have not really been explored. Because of this, I thought I’d just pick a random L. karasmontana gene sequence from the Kew Tree of Life dataset (https://treeoflife.kew.org/tree-of-life/6889) and have a bit of fun with it!
The gene I’ve picked (drumroll please) is… UVH6! More precisely, this is a Lithops ortholog of the UVH6 gene in Arabidopsis. There isn’t much we can infer about the Lithops gene with the information we have. All I can say is that there is a DNA sequence labelled “UVH6” in the Kew dataset, and that it is only a fragment of the total length of the Arabidopsis UVH6 gene (at 1002 bp long). However, this fragment shares 77% sequence identity with the equivalent region of the Arabidopsis gene, which is pretty good!

Figure 7. Left: A portion of the Blastn alignment of the putative UVH6 sequence from L. karasmontana (top) and the UVH6 sequence from Arabidopsis (bottom). (https://blast.ncbi.nlm.nih.gov/Blast.cgi). Right: Alphafold predicted structure of Arabidopsis UVH6 protein, because everyone likes pretty pictures (https://alphafold.ebi.ac.uk/entry/AF-Q8W4M7-F1).
Since the UVH6 gene in Lithops has not been studied, we can only infer its possible functions by looking at its homologs in other species, such as Arabidopsis. Based on its homology to well-characterized genes in yeast and humans, the Arabidopsis UVH6 probably codes for a helicase, which is an enzyme which is capable of separating the two strands which make up a DNA double helix [4]. The homologs of UVH6 in humans and in yeast have previously been shown to play important roles in repairing damaged DNA via a process called Nucleotide Excision Repair (NER) [4]. Incidentally, NER is particularly well-suited for repairing DNA lesions caused by exposure to UV radiation; It should come as no surprise, therefore, that Arabidopsis mutant plants which lack a functional UVH6 gene are sensitive (easily damaged) by exposure to UV [4]!
As I said before, we cannot say what the functions of UVH6 in Lithops may be; However, it would be relatively safe to assume that a plant living in regions which receive a lot of sunlight (e.g. lithops) would need mechanisms for DNA repair to prevent themselves from accumulating damage due to exposure to UV. Whether this resilience comes from UVH6 or something else, we cannot say… for now.
Works cited:
[1] Chial, H. (2008). DNA fingerprinting using amplified fragment length polymorphisms (AFLP): No genome sequence required. Nature education 1(1), 176.
[2] Field, K.J., George, R., Fearn, B., Quick, W.P., & Davey, M.P. (2013). Best of both worlds: Simultaneous High-Light and Shade-Tolerance Adaptations within Individual Leaves of the Living Stone Lithops aucampiae. PLoS One 8(10), e75671.
[3] Kellner, A., Ritz, C.M., Schlittenhardt, P., & Hellwig, F.H. (2011). Genetic differentiation in the genus Lithops L. (Rushioideae, Aizoaceae) reveals a high level of convergent evolution that reflects geographic distribution. Plant Biology 13(2), 368-380.
[4] Liu, Z., Hong, S., Escobar, M., Vierling, E., Mitchell, D.L., Mount, D.W., & Hall, J.D. (2003). Arabidopsis UVR6, a Homolog of Human XPD and Yeast RAD3 DNA Repair Genes, Functions in DNA Repair and is Essential for Plant Growth. Plant Physiology 132(3), 1405-1414.
[5] Loots, S., Nybom, H., Schwager, M., Sehic, J., & Ritz, C.M. (2019). Genetic variation among and within Lithops species in Namibia. Plant Systematics and Evolution 305, 985-999.
[6] Sajeva, M. & Oddo, E. (2007). Water Potential Gradients between Old and Developing Leaves in Lithops (Aizoaceae). Functional Plant Science and Biotechnology 1(2), 366-368.

Just a quick one this week!
I noticed these magnificent Jewelweeds (Impatiens capensis) while traveling a few weeks ago, and I just wanted to share them with you. They’re absolutely everywhere, and they have lovely flowers! They’re native to North America, and can generally be found in low-lying areas with wet soil.

Figure 1. Left: Patch of spotted jewelweed. Right: Close-up of the flowers.
The bees really seem to like them! However, getting to the nectar seems to be a bit of work; The bees have to dive deep into the flower to find it. As they do so, they brush up against the reproductive parts of the flower, ensuring pollination.

Figure 2. Left: Closeup of an Impatiens flower showing the location of the reproductive organs. Right: A bee inspecting and entering a flower to find nectar.
Fun! But what happens after the plant is done flowering is far more interesting. Jewelweeds are also known by another name – “touch-me-nots”. This is because the mature fruits explode at even the slightest touch! Actually, while I was working on this, I noted that this behavior is very similar to that of the Himalayan Balsam, which I have previously encountered while travelling. If you live in Europe or Asia, you may have seen them around. They have lovely purple flowers and explosive fruits. Anyhoo, I later found out that touch-me-nots and himalayan balsam are very closely related to each other, and are even in the same genus… so that makes sense. I feel pretty silly now, really. The explosive seed dispersal process has actually been studied in both species [1][3]. I’ll admit, both of these papers go way over my head – but it’s comforting to know that someone else has worked on this!
Right, it’s time for a demonstration. In Figure 3 below, I show an unexploded seed pod on the left, an exploded seed pod in the middle, and the seeds on the right.

Figure 3. Left/middle: Views of an Impatiens seed pod before and after the explosion. Right: Seeds!
As you can see, the walls of the fruit coil tightly during the explosion. This indicates that they are under a great deal of tension in the unexploded fruit. Deegan (2012) show that the fruit walls are held under tension by a membrane which connects them together [1]. The slightest tear in this membrane triggers a catastrophic failure in the structure [1].
How fast does this happen in our jewelweeds? Hayashi et al. (2009) report that the explosion takes approximately 4 milliseconds. I tried to film a seed pod using my phone, which can film at 120 fps. I got the following 4 consecutive frames:

Figure 4. Seed ejection sequence. These are consecutive frames from a 120 fps video. An ejected seed (marked with a red arrow) can be seen travelling away from the seed pod.
As you can see from Figure 4, the ejection begins in the second frame and is already complete by the third frame. The time between frames is ~8.33 ms, so our estimate roughly agrees with the measurement from Hayashi et al.
Alas, I have not been able to find much information about the genetics of explosive seed dispersal. So, I will leave you with this: A genome for Impatiens capensis has been published [2]! I’m sure some folks are going to have plenty of fun with this in the future.
Works cited:
[1] Deegan, R.D. (2012). Finessing the fracture energy barrier in ballistic seed dispersal. PNAS 109(14), 5166-5169.
[2] Gadagkar, S.R., Baeza, J.A., Buss, K., & Johnson, N. (2023). De-novo whole genome assembly assembly of the orange jewelweed, Impatiens capensis Meerb. (Balsaminaceae) using nanopore long-read sequencing. PeerJ 11, e16328.
[3] Hayashi, M., Feilich, K.L., & Ellerby, D.J. (2009). The mechanics of explosive seed dispersal in orange jewelweed (Impatiens capensis). Journal of Experimental Botany 60(7), 2045-2053.

Housekeeping!
I just wanted to apologize for missing last week’s regularly scheduled post. I have been travelling and did not have time to work on my blog. So… yeah, sorry about that. I’ll try to plan ahead better next time.
Before we start, I also wanted to let you know that I will be coming out with a new post every 2 weeks from now on, instead of every week. This is because I now have less free time than I did before (I got a job, lol). But don’t worry, we still have lots to talk about. Right then, to the post!
More about chamomile flowers than you ever needed/wanted to know:
I’ve been growing a chamomile plant in a little hydroponics unit sitting on a table indoors. It has been fun watching it grow, but recently I’ve become very interested in the flowers.
Like sunflowers, chamomile is a member of the Asteraceae. Members of this family have inflorescences – that is, clusters of many flowers – which together resemble a single flower. There are two kinds of flowers within the inflorescence: Ray flowers and disc flowers. In chamomile, ray flowers have large white petals and disc flowers have comparatively tiny petals. You can see an example of a chamomile inflorescence in Figure 1 below.

Figure 1. Chamomile inflorescence, showing the location of ray flowers and disc flowers.
You might be looking at this picture and thinking: “I don’t see any flower-shaped things in there. It just looks like one big flower.” That’s fair. So, let’s take a look at the ray and disc flowers under the microscope! Figure 2 shows a side view of a disc flower. The flower is so tiny that you can see the individual cells which make it up. We can see the petals, the stigma (a female reproductive part), and even scattered pollen grains.

Figure 2. A chamomile disc flower at 100X magnification and blown up further to a larger size.
In Figure 3, we can see a ray flower. The structure of the female reproductive organs is similar to the disc flower; The stigma can be seen here, too. However, chamomile ray flowers are pistillate, meaning that they lack the male reproductive structures [6]. Neat! Furthermore, the petals are fused into a single large, flat plane that is comparatively enormous in size (I had to cut it off to fit the flower under the microscope).

Figure 3. A chamomile ray flower (with petals cut off) at 100X magnification. The fused petals are enormous and would have extended a long way to the right, had they not been severed.
Check this out!
This is all very fascinating, of course. But the REALLY cool thing about chamomile flowers is that they can move!
No, really! I’m not crazy…. They can move – just very slowly. Now, I’ve been growing my chamomile plant in day-night light cycles. During the day, the petals of the ray flowers are erect and point out sideways. At night, the petals of the ray flowers hang downwards. To illustrate this, I photographed the same 2 flowers over a couple days to show this movement in action – see Figure 4 below.

Figure 4. Daily petal movements of chamomile ray flowers. This image series follows the same pair of flowers across 2 days.
While petal movements in chamomile are certainly striking, I should point out that daily rhythmic movements are actually quite common in plants. For example, if you’ve ever kept Oxalis triangularis (a common houseplant), you might also notice that the leaves and flowers move up and down, depending on the time of day (See Figure 5 below). Similarly, the leaves of the model plant Arabidopsis thaliana also move up and down depending on the time of day [2], though this movement is somewhat less striking than in Oxalis and involves a different organ (the petioles as opposed to pulvini within leaves themselves).

Figure 5. Daily movements in an Oxalis kept as a houseplant. Left: The plant during the day. Right: The plant at night. These are my photos, but not my plant.
In Oxalis and Arabidopsis, these rhythmic movements are controlled by the circadian clock [2][3]. The circadian clock is an intrinsic biological rhythm which controls daily biological cycles. You have a circadian clock which controls your sleep patterns (this is why you sleep at roughly the same time each day) and various aspects of your metabolism. Plants also have a circadian clock which controls rhythmic movements, among other things.
Time to get a new watch…
One defining characteristic of circadian rhythmics is that they persist when organisms are moved from cycling to constant conditions [3]. For example, when Arabidopsis is shifted from day/night light cycles to constant light conditions, up-and-down leaf movements continue on a roughly 24-hour cycle [2]. With this in mind, I asked myself whether the flower movements in chamomile were also controlled by the circadian clock.
To test this, I severed some chamomile inflorescences, gave them some water, and put them into constant dark conditions (in my closet). I harvested two inflorescences at 4 PM (daytime), which roughly corresponds to the time when the ray flower petals are most erect. I also harvested two inflorescences at midnight. At this time, the ray flower petals hang loosely. (For your reference, my lights are kept on from 6 AM to 11 PM). Then, I tracked the movement of the flower petals over the next couple of days.
Hypothesis: If the flower movements are controlled by circadian rhythms, then the petals should continue to move up and down in constant dark conditions.
Great! We’re all set. So… what actually happened? The results actually did not support my hypothesis. As you can see in Figure 6, the inflorescences harvested during the day drooped after being moved to constant dark, and did not re-open. Meanwhile, the inflorescences harvested at midnight remained drooped throughout the entire experiment.

Figure 6. Petal movements in chamomile flowers kept in constant darkness. The top row of images shows flowers harvested at 4 PM, during the day. The bottom row of images shows flowers harvested at midnight. Images in each row are in chronological order from left to right.
Why did this happen? I don’t know, but I’ve thought of a couple possible explanations. Let’s discuss.
POSSIBLE EXPLANATION 1: Flower movements in chamomile are not controlled by the circadian rhythm, but are rather directly controlled by present light levels.
I think this is unlikely for a couple of reasons. First, I observed that the petals on my chamomile plant tend to start drooping at the end of the day, BEFORE the lights go off. In other words, they are anticipating an impending change in light conditions. This would not happen if petal movements were determined by light alone.
I also tested this theory directly. To do this, I took my chamomile inflorescences that had been sitting in the dark for a few days (and were super droopy) and moved them back into the light. If light is necessary and sufficient to cause petals to stand up, then I would expect the petals in my cut inflorescences to do just that. They appeared to make what seemed to be a half-hearted attempt to stand up after many hours of light exposure, but ultimately did not move much. Interesting.
POSSIBLE EXPLANATION 2: The act of severing inflorescences from the rest of the chamomile plant results in a loss of circadian petal movement. I can think of two possible reasons for this. One possibility is that petal movements are controlled remotely by a circadian oscillator in another part of the plant (the leaves, for instance). Another possibility is that petal movement requires a specific nutrient, which severed flowers in water would be unable to acquire.
Sadly, I still haven’t had the time to test these ideas. This is because I cut off all of my chamomile flowers for my previous experiment (and I didn’t have many to begin with!). However, when my plant recovers and generates more flowers, I should be able to do a small experiment. If rhythmic petal movements require that the inflorescence be attached to the rest of the plant, then these movements should persist if the whole plant is transferred to constant dark conditions. I’ll let you know when the results of that test become available.
Gene of the week:
Tragically, I was not able to find any research about the genetics underpinning flower petal movements in chamomile or in a closely related species. Therefore, I have been forced to choose a Gene of the Week which is only tangentially related to what we were talking about. I still think you’re going to like it, though.
This week’s Gene of the Week is ELF3. Funny name, eh? ELF3 stands for “EARLY FLOWERING 3”, because Arabidopsis plants lacking a functional copy of this gene flower early [4]. … I guess that makes sense. The ELF3 protein is an important component of the circadian clock in Arabidopsis. Without it, plant movements and other circadian-regulated processes are arrhythmic in constant light [5]. Interestingly however, elf3 mutant Arabidopsis plants still maintain rhythmicity in constant dark conditions [5].
What does the ELF3 protein do? Famously, it is part of a larger protein complex (called the Evening Complex) which controls the expression of other genes [8]. As its name suggests, the Evening Complex is most active in the evening. Very generally, the Evening Complex helps to ensure that daily oscillations in gene expression occur at the correct time. Interestingly, recent research has also shown that ELF3 also has functions independently of the evening complex [7]… so it does rather a lot!
Now, the BIG question: Does chamomile have an ELF3 gene? Well, my favorite ortholog database (OMA: https://omabrowser.org/oma/home/) does not include chamomile. And I wasn’t able to find a proteome for chamomile, either. And a Blast search (think search engine, but for DNA/protein sequences) using the Arabidopsis ELF3 DNA sequence as a query did not find an ELF3 DNA sequence in chamomile. What to do?
Fortunately, there is a reference genome for chamomile which is partially annotated [1]! The authors of this paper identified a probable ELF3 ortholog, which is noted in their supplemental data. If we take the first exon in this gene and translate it into a predicted protein sequence (beginning with the predicted start codon), and compare it to the Arabidopsis ELF3 protein sequence, we find that the two are very similar! (Figure 7). So it seems chamomile has an ELF3 ortholog after all.

Figure 7: Alignment of the beginning of the Arabidopsis and chamomile ELF3 proteins (chamomile is a hypothetical protein sequence).
Works cited:
[1] Cho, W., Feng, J., Knauft, M., Albrecht, S., Himmelbach, A., Otto, L., & Mescher, M. (2025). An annotated haplotype-resolved genome sequence assembly of diploid German chamomile, Matricaria chamomilla. Scientific Data 12, 358.
[2] Dornbusch, T., Michaud, O., Xenarios, I., & Fankhauser, C. (2014). Differentially Phased Leaf Growth and Movements in Arabidopsis Depend on Coordinated Circadian and Light Regulation. The Plant Cell 26(10), 3911-3921.
[3] Edery, I. (2000) Circadian rhythms in a nutshell. Physiological Genomics 3, 59-74.
[4] Hicks, K.A., Albertson, T.M., & Wagner, D.R. (2001). EARLY FLOWERING 3 Encodes a Novel Protein that Regulates Circadian Clock Function and Flowering in Arabidopsis. The Plant Cell 13(6), 1281-1292.
[5] Hicks, K.A., Millar, A.J., Carré, I.A., Somers, D.E., Straume, M., Meeks-Wagner, D.R., & Kay, S.A. (1996). Conditional circadian dysfunction of the Arabidopsis early flowering 3 mutant. Science 274(5288), 790-792.
[6] Matricaria chamomilla (Wild chamomile, German chamomile).Retrieved September 16, 2025, from: https://wwv.inhs.illinois.edu/data/plantdb/detail/2185
[7] Nieto, C., López-Salmerón, V., Davière, J., & Prat, S. (2015). ELF3-PIF4 interaction regulates plant growth independently of the Evening Complex. Current Biology 25(20, 187-193.
[8] Nusinow, D.A., Helfer, A., Hamilton, E.E., King, J.J., Imaizumi, T., Schultz, T.F., Farré, E.M., & Kay, S.A. (2011). The ELF4-ELF3-LUX complex links the circadian clock to diurnal control of hypocotyl growth. Nature 475(7356), 398-402.
[9] You, L., Tuo, W., Dai, Z., Wang, H., Ahmad, S., Peng, D., & Wu, Shasha. (2023). Effects of light intensity, temperature, and circadian clock on the nyctinastic movement of Oxalis triangularis ‘Purpurea’. Technology in Horticulture 3, 11.

I regret to inform you that preparation for this week’s blog post has taken me longer than I expected. Don’t worry, that post will come out next week. While I’m busy cooking that up, I would just like to share with you a little inspirational story from the garden.
I’ve grown a bunch of sunflowers this season. They’ve done really well in pots, but the ones that I planted in the ground have struggled. When I sowed directly, chipmunks ate the seeds. When the seedlings sprouted, they were either pulled out by birds or eaten by slugs, no matter how much repellent I used or how many seeds I planted.
Therefore, I tried transplanting larger plants into the ground. At this stage of their growth, sunflowers really don’t seem to like being transplanted. Even so, a few plants got to a decent size – before the deer ate their heads off. Despite these hardships, one singular plant managed to flower and even to set seed…

And here it is! This particular specimen is an example of a “Titan” sunflower. With this name in mind, it is apparent that this plant has had a really tough life. But in the end, it did the best with what it had, and still made a beautiful flower. Just something to think about.

I’ll be the first to admit that this title doesn’t make much sense. We’ll only be talking about one kind of plastid today: the etioplast! Etioplasts are plastids which inhabit plant tissues grown in the dark. They undergo rapid conversion to chloroplasts when light is available to power photosynthesis.
One of the easiest ways to “make” etioplasts at home is to grow seedlings in the dark. In Figure 1, you can see a sunflower seedling grown in the dark, side-by-side with seedlings grown in the light. These seedlings are all the same age, but there are some notable differences! For starters, the dark-grown seedling is a lot smaller. Furthermore, if we zoom in and look at the cotyledons (embryonic structures resembling leaves), we can see that they are yellow in the dark-grown seedling and green in the light-grown seedlings. This yellow coloration indicates the presence of etioplasts!

Figure 1. Left: Dark-grown sunflower seedling (in my hand) compared to light-grown sunflower seedlings of the same age. Right: A close-up of the cotyledons of the dark-grown seedling. They contain lots of etioplasts!
Ok, great! Can we see some etioplasts under the microscope?
… *sigh*. I tried. I really did. But I encountered some difficulties in doing so. The main problem is that the cotyledon cells in the dark-grown seedling are very small and densely packed together. Light attenuates quickly in such densely-packed tissue, making it difficult to visualize on the microscope (I just see dark-colored blobs). Fine. So next, I tried squishing the tissue in the hopes of splitting a few individual cells off to look at. But I found that the cells were very… crumbly. When I squished the tissue, the cells just ruptured and released their contents in a giant maelstrom of crap (See Figure 2 below). Some of the little globules you can see are probably etioplasts, but since they are not darkly pigmented, it is impossible to differentiate them from other cell contents.

Figure 2. The big mess made by squishing tissue from dark-grown cotyledons.
One possible explanation for the brittle-ness of the dark-grown cotyledon cells could be that their cell walls are thinner than in light-grown seedlings. I couldn’t find information about cotyledons specifically, but I did find this source which shows that cell wall thickness increases in the hypocotyls (stems)of sunflower seedlings after they are exposed to light [2]. This makes intuitive sense, since plants with access to light can photosynthesize and obtain more carbon to build cell walls with.
Wherefore art thou, chloroplast?
OK, so we can’t easily look at etioplasts directly. However, we can observe the etioplast-to-chloroplast transition by looking at whole seedlings! As I mentioned before, etioplasts become chloroplasts when they are exposed to light. We can see this change by looking at the color change of dark-grown cotyledons, from yellow to green! For an example, see Figure 3 below. This color change occurs because light stimulates production of chlorophyll, the green pigment required for photosynthesis.

Figure 3. Dark-grown seedlings vs. two dissected cotyledons from a light-grown seedling. Top: The dark-grown seedlings were just transferred to light. Bottom: The dark-grown seedlings were exposed to light for 8 hours.
How do etioplasts sense light, and how does this lead to chlorophyll production? I’m glad you asked! Etioplasts contain an important enzyme called protochlorophyllide oxidoreductase (POR for short) [3]. This enzyme catalyzes a key step in chlorophyll biosynthesis. I was surprised to learn that the enzyme itself is light-activated – no other upstream signaling mechanisms are needed [3]! Very cool.
What would happen if a plant were deficient in the POR enzyme? As you might expect, Arabidopsis mutants with reduced levels of POR have reduced chlorophyll content and various other chloroplast defects [1]. Arabidopsis actually has 3 POR genes. Single mutants don’t have an obvious phenotype, but double mutants do [1]. The presence of multiple genes which can perform the same tasks is known as genetic redundancy. In the case of POR, the porB porC double mutant is seedling lethal – that is, plants cannot grow beyond the seedling stage before dying [1].
Mixed results:
We’re all scientists here. And scientists measure things! So…. Is there a way to quantify the greening process?
Well, sure. You could extract and quantify the concentration of chlorophyll in seedlings – but this requires a bunch of specialized equipment that I don’t have. However, I did come across this interesting paper which proposes a way to estimate chlorophyll content from RGB photographs, such as those you can take with a smartphone camera [4]! In particular, they found that the ratio of green to red pixel channel values was a reasonably good proxy for chlorophyll content [4]. This makes intuitive sense if you consider that in RGB, green color is created by having a high green channel value, while yellow is made by mixing red and green channels together. A low G:R ratio gives yellow and indicates low chlorophyll content, while a high G:R ratio gives green and indicates a high chlorophyll content!
With this in mind, I photographed cotyledons from 3 different dark-grown seedlings after they were exposed to light, and quantified the G:R ratio of the pixels making up these cotyledons using an image-processing software called FIJI. Below, you can see that the G:R ratio for my three seedlings increases with time as they were exposed to light, indicating accumulation of chlorophyll. It works!
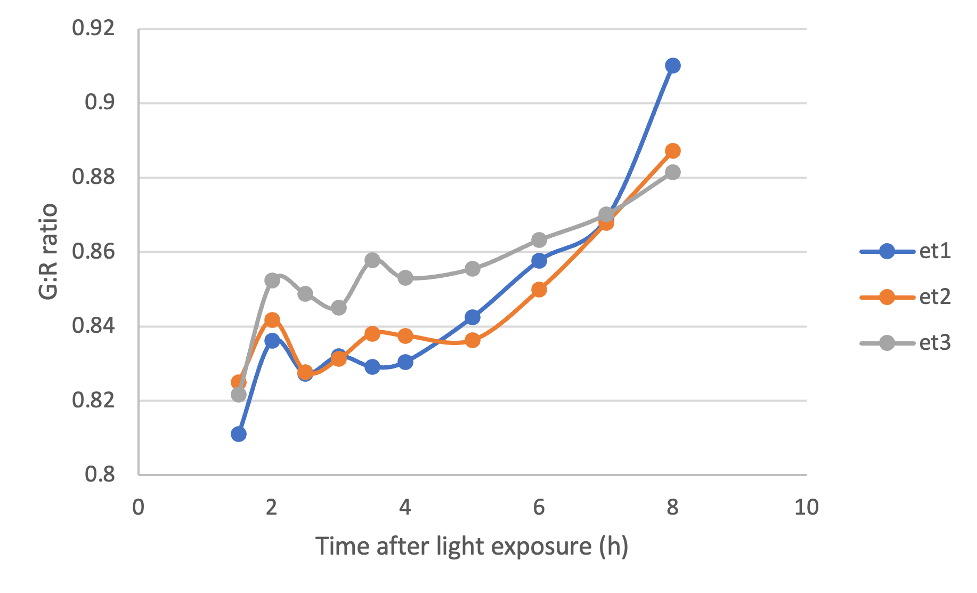
Figure 4. G:R ratio of rectangular selections of cotyledons from 3 dark-grown seedlings over time after they were exposed to light.
It… er… might have been a bit too early to celebrate. I also took the G:R ratio of a couple of light-grown cotyledons. We would expect these values to be relatively constant over time, since the chlorophyll content of mature cotyledons shouldn’t change meaningfully over only a few hours. However, if we plot the G:R ratio of light-grown cotyledons on the same graph as our dark-grown cotyledons, we get something that looks like Figure 5 below.

Figure 5. G:R ratio of rectangular selections of cotyledons from 3 dark-grown seedlings and 2 light-grown cotyledons over time.
What to heck?! The G:R ratios of the light-grown cotyledons are super variable! But why? Troubleshooting is an important part of the scientific process, so let’s think of some possibilities.
One issue that I found was that the surfaces of the light-grown cotyledons were somewhat reflective under my lights, especially when water droplets were present on their surface. Why were there water droplets? Since the cotyledons no longer had access to water, I was misting them with water constantly to keep them moist and alive. Unfortunately, this probably also had an effect on my photographs.
Another interesting thing that I noticed was that the blue channel values for pixels from the dark-grown cotyledons were virtually 0 throughout the entire experiment. However, the blue channel values for pixels from the light-grown cotyledons were higher and quite variable. In instances where the blue channel values were high, the green value channels tended to be lower, indicating a hidden relationship between the blue and green channels. It is possible that this “hidden relationship” made my results more variable. I don’t know exactly why this occurred. One interesting possibility is that this is due to the presence of other pigments (such as anthocyanins) in the light-grown seedlings, but I cannot say for certain.
I’m definitely going to try this again, with an improved protocol. Stay tuned.
Gene of the week:
Well alrighty, it’s that time again! This week’s genes of the week will be the POR genes. As previously mentioned, Arabidopsis has 3 POR genes, which act redundantly with each other to catalyze chlorophyll production in etioplasts undergoing the transition to chloroplasts [1].
All of the POR proteins in Arabidopsis are roughly 400 amino acids long and have a mass of around 43 kDa (according to their respective UniProt entries). If we align their sequences using Clustal Omega (https://www.ebi.ac.uk/jdispatcher/msa/clustalo), we can see that their sequences are extremely similar (See Figure 6) – indicating that the 3 genes probably emerged from a recent duplication event.
But it gets crazier! I found this interesting paper which uses a technique called cryo-electron microscopy to look at the structure of POR [5]. They found that POR proteins come together to form enormous tubular structures! Apparently, these tubes help to organize internal membranes within the plastid [5]. And they look oh-so-cool! See Figure 6 below for an example of the structure.

Figure 6. Left: Sequence alignment of the 3 POR proteins from Arabidopsis. Right: Structure of PORB. The sequences and the structure were obtained from the following UniProt entries:
https://www.uniprot.org/uniprotkb/Q42536/entry#sequences
https://www.uniprot.org/uniprotkb/P21218/entry#sequences
https://www.uniprot.org/uniprotkb/O48741/entry#sequences
Works Cited:
[1] Frick, G., Su, Q., Apel, K., & Armstrong, G.A. (2003). An Arabidopsis porB porC double mutant lacking light-dependent NADPH:protochlorophyllide oxidoreductases B and C is highly chlorophyll-deficient and developmentally arrested. The Plant Journal 35(20, 141-153.
[2] Kutschera, U. (1990). Cell-wall synthesis and elongation in the hypocotyls of Helianthus annuus L. Planta 181, 316-323.
[3] Menon, B.R.K., Davidson, P.A., Hunter, C.N., Scrutton, N.S., & Heyes, D.J. (2009). Mutagenesis Alters the Catalytic Mechanism of the Light-driven Protochlorophyllide Oxidoreductase. Journal of Biological Chemistry 285(3), 2113-2119.
[4] Nasoha, N.Z., Ibrahim, N.U.A., Harith, H.H., Jamaludin, D., & Abd Aziz, S. (2025). Linear regression and machine learning modelling for chlorophyll content estimation using leaf red, green, and blue images. Food Research 9(1), 94-100.
[5] Nguyen, H.C., Melo, A.A., Kruk, J., Frost, A., & Gabruk, M. (2021). Photocatalytic LPOR forms helical lattices that shape membranes for chlorophyll synthesis. Nature Plants 7, 437-444.

How do plants sense gravity?
I’ve been growing some sunflower seedlings indoors under artificial lights. To demonstrate that plants can sense gravity, I turned one pot on its side and left it in a dark cupboard overnight. As you can see in Figure 1, the seedlings reorient their growth so that they once again face upward. But how do they know which way the gravity is going??
The answer is specialized plastids called amyloplasts, which contain large quantities of starch (“amlyo” is Greek for starch). We covered the concept of plastids in last week’s post. In short, plastids are organelles (subcellular structures) which have their own DNA and which live inside of plant cells. They can take on a number of different identities and may become chloroplasts, chromoplasts, amyloplasts, etc. Plants sense gravity using specialized amyloplasts called statoliths (Greek for “standing stone”, because they look like little stones! You’ll see why in a moment).

Figure 1. Left: sunflower seedlings used to test gravity-sensing. Right top: The same pot turned on its side. Right bottom: the same pot after sitting on its side in the dark overnight. One seedling was removed for microscopy (you’ll see why in a moment!)
Let’s see some statoliths!
Okey dokey! Let’s see some statoliths! But how?
We’ll need to look at cells in the hypocotyl of our sunflower seedlings. The hypocotyl is the stem of the seedling (See Figure 2 left), which is derived from an embryonic structure that forms early in seedling development. Note that roots also have statoliths and can sense gravity independently of the stem… but I will discuss those in a later blog post.
Anyway, statoliths in the model plant Arabidopsis thaliana are known to be found in the endodermal cell layer in stems [7]. Before going any further, I wanted to check whether the same was true for sunflower hypocotyls. To do this, I took a horizontal section through the hypocotyl (See Figure 2 right) and had a look under the microscope.

Figure 2. Left: Sunflower seedlings, showing the location of the hypocotyl and cotyledons (leaf-like structures).
The endodermis is a layer of cells that surrounds the pith and vascular tissue. In an unstained section of hypocotyl, it is extremely difficult to make out (See Figure 3 top left). Fortunately, we have some Lugol’s iodine at our disposal! You might remember from a science class at some point that iodine stains starch. Using Lugol’s iodine to stain our hypocotyl sections allows us to see the location of our statoliths – and by extension, the endodermis. You can see a hypocotyl section stained with Lugol’s iodine in the top right of Figure 3. A thin line of black spots belies the position of the endodermis. If you look even more closely, you can see individual statoliths within the endodermal cells (Figure 3 bottom).

Figure 3. Top left: Unstained section of sunflower hypocotyl at 100X magnification. Top right: Section of sunflower hypocotyl stained with Lugol’s iodine at 100X magnification. The position of the endodermis is indicated. Bottom: Endodermis and surrounding tissues at 400X magnification. Individual statoliths are visible.
Statoliths facilitate gravity sensing because they are denser than other cell contents and sink to the bottom of cells [7]. More on that in a minute. But this means that if we want to see statoliths in action, we need to take a vertical section of hypocotyl and view cells from the side. (See Figure 4 top for the location of the cuts I made). When we look at these vertical sections without staining, it is difficult to identify the endodermis cells (Figure 4 bottom left). However, after staining with Lugol’s iodine, the location of the statoliths/endodermis becomes clear (Figure 4 bottom right)!

Figure 4. Top: Locations of cuts to make to take vertical sections of hypocotyl tissue. Bottom left: Unstained hypocotyl section, with visible tissues labelled. Bottom right: Hypocotyl section stained with Lugol’s iodine. The endodermis, which contains statoliths, is visible.
Statolith sedimentation:
As previously mentioned, statoliths are believed to facilitate gravity sensing because they sink to the bottom of the cells. Sinking of the statoliths sets off a signaling cascade which ultimately results in a redistribution of the growth hormone auxin within the stem/hypocotyl, which leads to asymmetric cell elongation, which in turn allows plants to change the direction of their growth [5][6]. Exactly how the statolith sensing mechanism works is still being intensively studied. The review article by Kawamoto & Morita [4] provides an excellent summary of several lines of research, but we still don’t fully understand how we get from statoliths sinking –> auxin redistribution.
Er… right. Given that we don’t understand precisely how statolith-mediated gravity sensing works, how do we even know that these statoliths are important at all?? Fortunately, there is good evidence that points in this direction. For example, Arabidopsis mutants which lack the endodermal cell layer entirely are agravitropic (do not respond to gravity) [3]. Furthermore, Arabidopsis mutants which lack starch-filled amyloplasts, such as pgm (phosphoglucomutase) mutants, have a weaker gravitropic response (though it is not entirely gone) [2].
All very cool. But can we see statolith sedimentation for ourselves? Sure we can! First, I looked at statoliths in a vertical section of hypocotyl in a seedling that was growing vertically upwards. You can see that in Figure 5 that the statoliths accumulate on the bottom of the cells, as expected. Since the cell boundaries are difficult to see, I’ve highlighted them in red.
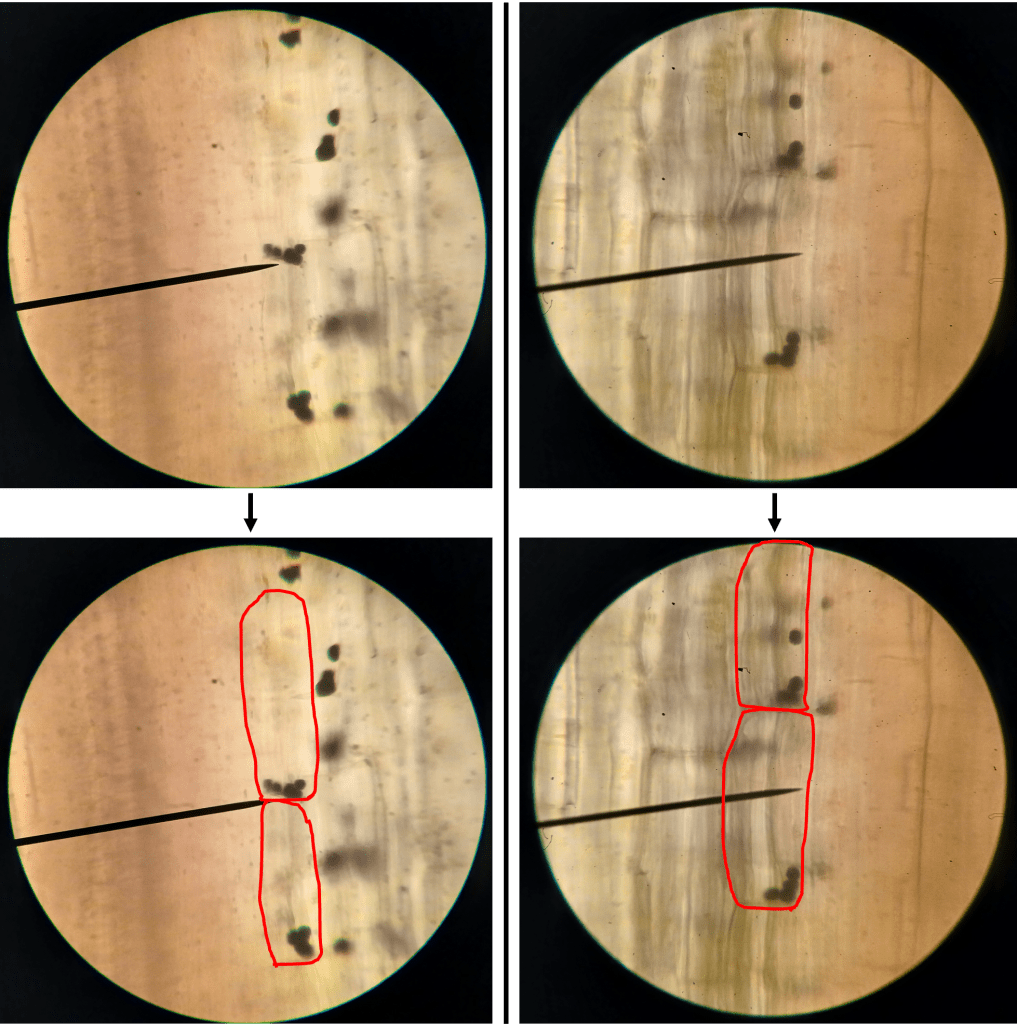
Figure 5. Statoliths in sunflower hypocotyls in a seedling which was growing vertically. Top: Unaltered images. Bottom: Images with cell boundaries highlighted for clarity.
Very good. Now, what happens if we tilt a seedling onto its side, wait a few hours, and then look at the statoliths? See for yourself in Figure 6! Here, statoliths accumulate on the lower side of the cells, as expected.

Figure 6. Statoliths in sunflower hypocotyls in a seedling which was tilted horizontally for several hours. Top: Unaltered images. Bottom: Images with cell boundaries highlighted for clarity.
Gene of the week:
And just like that, it’s time for gene of the week! This week’s gene will be PGM (phosphoglucomutase) in Arabidopsis. PGM codes for an enzyme which is essential for the biosynthesis of starch in amyloplasts. As previously noted, pgm mutants have a reduced gravitropic response [2]. As expected, the UniProt database entry (https://www.uniprot.org/uniprotkb/Q9SCY0/entry#subcellular_location) notes that the PGM protein localizes to plastids. It also tells us that the PGM gene in Arabidopsis is located at the AT5G51820 locus on chromosome 5, and codes for a protein which is 623 amino acids long, with a mass of ~68 kDa. There is a nice alphafold structure available:
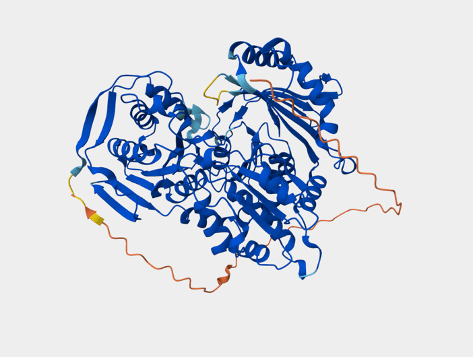
Figure 7. Alphafold structure of Arabidopsis PGM, taken from its UniProt entry.
I did a quick search of the OMA database (https://omabrowser.org/oma/home/) to see if there are any known orthologs of Arabidopsis PGM in sunflowers. For genes involved in such essential biochemical pathways, I would expect to see lots of orthologs with a high degree of sequence similarity across many species, so this should be a breeze. Indeed, OMA tells us that a predicted sunflower protein called “HELAN14524” exists, which has a similar sequence to Arabidopsis PGM. Furthermore, it is possible to predict that the sunflower PGM protein also localizes to plastids, because it contains a Transit Peptide sequence – a protein sequence motif that acts kind of like a “mailing address” to send proteins to plastids [1].
Works cited:
[1] Bruce, B.D. (2000). Chloroplast transit peptides: structure, function, and evolution. Trends in Cell Biology 10(10), 440-447.
[2] Caspar, T., & Pickard, B.G. (1989). Gravitropism in a starchless mutant of Arabidopsis. Planta 177, 185-197.
[3] Fukaki, H., Wysocka-Diller, J., Kato, T., Fujisawa, H., Benfey, P.N., & Tasaka, M. (2002). Genetic evidence that the endodermis is essential for shoot gravitropism in Arabidopsis thaliana. The Plant Journal 14(4), 425-430.
[4] Kawamoto, N., & Morita, M.T. (2022). Gravity sensing and responses in the coordination of the shoot gravitropic setpoint angle. New Phytologist 236, 1637-1654.
[5] Rakusová, H., Abbas, M., Han, H., Song, S., Robert, H.S., & Friml, J. (2016). Termination of Shoot Gravitropic Responses by Auxin Feedback on PIN3 Polarity. Current Biology 26(22), 3026-3032.
[6] Wang, X., Yu, R., Wang, J., Lin, Z., Han, X., Deng, Z., Fan, L., He, H., Deng, Z.W., & Chen, H. (2020). The Asymmetric Expression of SAUR genes Mediated by ARF7/19 Promotes the Gravitropism and Phototropism of Plant Hypocotyls. Cell Reports 31(2), 107529.
[7] Wyatt, S.E., Rashotte, A.M., Shipp, M.J., Robertson, D., & Muday, G.K. (2002). Mutations in the Gravity Persistence Signal Loci in Arabidopsis Disrupt the Perception and/or Signal Transduction of Gravitropic Stimuli. Plant Physiology 130(3), 1426-1435.

What are plastids?
You’ve almost certainly heard about chloroplasts, which are the compartments inside plant cells that perform photosynthesis. They’re small, green, and plant cells have lots of them! Well, in tissues that lie above ground, anyway. Chloroplasts look like this:

Figure 1: Chloroplasts inside tomato pericarp cells at 400X magnification (and further enlarged).
What if I told you that chloroplasts can transform? Chloroplasts are a kind of plastid, and plastids can take several different forms, depending on the cell type and the situation. In ripening fruits of the Solanaceae (including peppers and tomatoes), chloroplasts can become chromoplasts – structures which produce pigments that give the fruit color [8]. More on those in a bit.
What exactly are plastids? Plastids are specialized, membrane-bound organelles (subcellular structures) which carry out a variety of functions in plant cells. Plastids are notable because they have their own genome (DNA), which is separate from the main genome found in the nucleus of the plant cell [8]. This is because plastids are likely the result of endosymbiosis – they were once free-living organisms that were swallowed up by a larger cell, decided they liked it in there, and decided to set up shop [11]. Mitochondria are believed to have originated in a similar way. In fact, your mitochondria (along with the mitochondria of all plants) probably originated from the same endosymbiosis event [6]! It’s a shame humans didn’t get plastids, really. That would have been fun. Oh well…
Tomato chromoplasts:
Right, so I mentioned that tomato chloroplasts turn into chromoplasts during the ripening process. I just had to see this happening for myself! You too can do this at home if you have a basic light microscope that can take you to around 400X magnification. Seeing plastids with this equipment can be a little finicky. The trick is to take tissue samples that are as thin as possible – preferably only 1 cell thick. This is because plant tissue rapidly dims light from the microscope (due in part to the presence of plastids!) and scatters the light, making it hard to resolve a clear image. To take my pictures, I took the tiniest piece of pericarp (tissue under the epidermis) I could separate, and gently squished it between the slide and the coverslip to separate cells from each other. It was messy and annoying… but it worked in the end!
In the top left of Figure 2 below, you can see the tomatoes I sampled from. (In case you’re curious, the tomato variety used here is Black Opal, a kind of cherry tomato). The green tomato on the top is unripe, and the red tomato on the bottom is ripe and ready-to-eat! I began by looking for chloroplasts in the unripe tomato. You can see my whole field of view (Figure 2 top right), and a blown-up part of the same image (Figure 2 bottom). The chloroplasts are the little green blobby things. You can also see from this image that chloroplasts tend to accumulate near the edges of cells.

Figure 2. The image in the top left shows the tomatoes used for sampling. The top right shows pericarp cells from the unripe tomato at 400X magnification. The bottom image is a blown-up version of the same image, which clearly shows the chloroplasts.
Okay, great! Now let’s look for the chromoplasts in the ripe tomato. Using the same sample preparation method, I was able to isolate a few pericarp cells from the ripe tomato and image them. If you look at Figure 3, the chromoplasts are the small red structures you can see inside the cell.
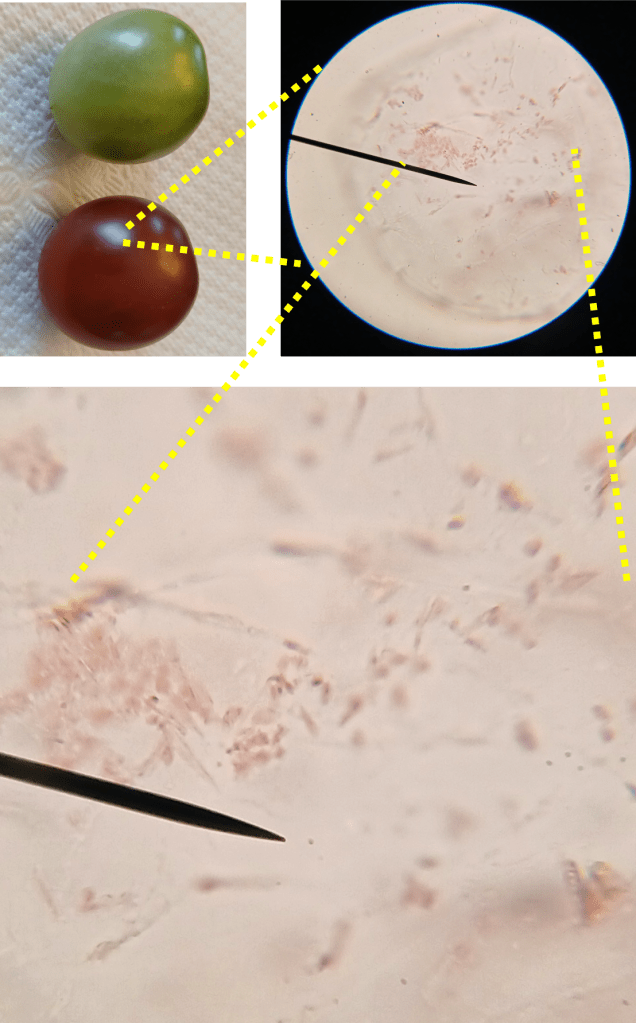
Figure 3. The image in the top left shows the tomatoes used for sampling. The top right shows pericarp cells from the ripe tomato at 400X magnification. The bottom image is a blown-up version of the same image, which clearly shows the chromoplasts.
Success! (I can’t believe that worked). To learn a bit more about tomato chromoplasts, let’s do a quick bit of Q and A.
1. Why are the chromoplasts red?
This color change occurs because chromoplasts accumulate various carotenoid pigments, including lycopene [12]!
2. Why are the chromoplasts all… spiky looking?
Great observation! This is because carotenoid pigments form long, crystal-like aggregates within the chromoplast [9]. Even though they may experience a dramatic increase in length, chromoplasts manage to maintain their membrane around the growing crystals [7].
3. Do chloroplasts experience changes in gene expression as they transform into chromoplasts?
Yes! One transcriptomic study showed that the expression of most plastid genes is downregulated as chloroplasts become chromoplasts, with the exception of a gene called accD [4]. The accD gene is involved in fatty acid synthesis and might be important for producing new membrane to encapsulate growing carotenoid crystals [4].
4. Do we know about other genes which are important for the chloroplast-chromoplast transition?
Yes! One interesting tomato mutant is the so-called “green flesh” mutant, which exhibits plastids that are apparently unable to complete the chloroplast-chromoplast transition [2]. The mutation which causes the green flesh phenotype was narrowed down to a gene called “STAY-GREEN” (SGR) in tomato [1]. Mutations in an ortholog of this gene in pepper cause a similar phenotype [1]. An ortholog has also been identified in the model plant Arabidopsis, though Arabidopsis does not possess chromoplasts [10]. Remember, orthologs are genes which share a common ancestor, but are found in separate species.
Now that we know a little bit more about tomato chromoplasts, I’d just like to wrap up this section with some bonus images. Figure 4 is an image of some cells I found which contain both chromoplasts AND chloroplasts. It is evidently mid-transition. Pretty neat!
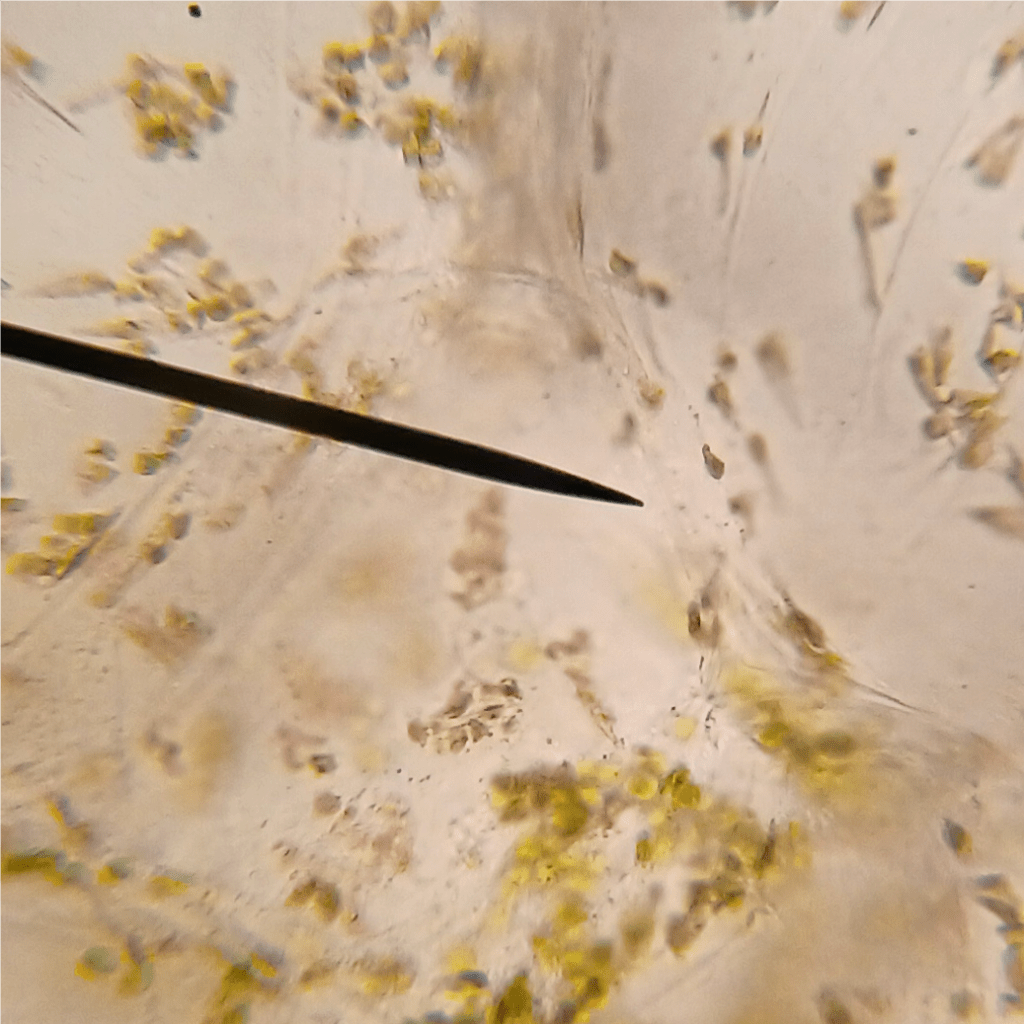
Figure 4. Tomato pericarp cell containing both chloroplasts and chromoplasts.
Figure 5 is an image of tomato epidermal (skin) cells. As far as I can tell, these don’t really have any color at all. But man, look at those extra thick cell walls! If I had to make an uneducated guess, I’d say that these cell walls are the reason the skin is mechanically tougher than the squishy inside of the fruit.

Figure 5. Tomato epidermis at 400X magnification.
Pepper chromoplasts:
I’ve mentioned that peppers also possess chromoplasts in ripe fruit. I just so happen to have a couple pepper plants growing out on the deck, so I figured it wouldn’t hurt to have a look! Figure 6 shows my attempt to view chromoplasts in the pericarp cells of a Hot Red Cherry pepper. Not too shabby.

Figure 6. The image in the top left shows the peppers used for sampling. The top right shows pericarp cells from the ripe pepper at 400X magnification. The bottom image is a blown-up version of the same image, which clearly shows the chromoplasts.
Like tomato chromoplasts, pepper chromoplasts also contain high levels of carotenoid pigments, which give them their red color [5]. And just as in tomato, most plastid genes are downregulated during the chloroplast-chromoplast transition, except for accD [5]. Neat! You can see that the shape of these chromoplasts is a bit different, however. They’re more globular, without the crystal-like structures visible in tomato chromoplasts.
Gene of the week!
Well, that was fun! Er… I enjoyed myself anyway. All right then, here’s the gene of the week: SGR1 (STAY-GREEN 1)! I mentioned earlier that this gene is required to complete the chloroplast-chromoplast transition in tomatoes. How does it work?
I couldn’t find much info about the molecular function of SGR1 in tomatoes. However, an ortholog in Arabidopsis is known (See sequence alignment in Figure 7 top left)! In Arabidopsis, the SGR1 gene codes for a protein which functions as an enzyme called Magnesium Dechelatase [1]. This enzyme is important for the degradation of chlorophyll, as it removes the coordinated Magnesium atom from within the porphyrin ring of the molecule (Figure 7 top right). Chlorophyll degradation is of course important as chloroplasts transition to chromoplasts and lose their green coloration.
A predicted structure for the SGR1 protein is available for both tomato and Arabidopsis. They both look pretty similar, but this is unsurprising given the high sequence similarity between the two proteins. It is important to note that SRG1 in its native form might exist as an oligomeric complex – that is, multiple copies of the protein may interact and form a larger structure [3]. According to the UniProt database, the tomato protein is 272 amino acids long and has a mass of roughly 30 kDa.
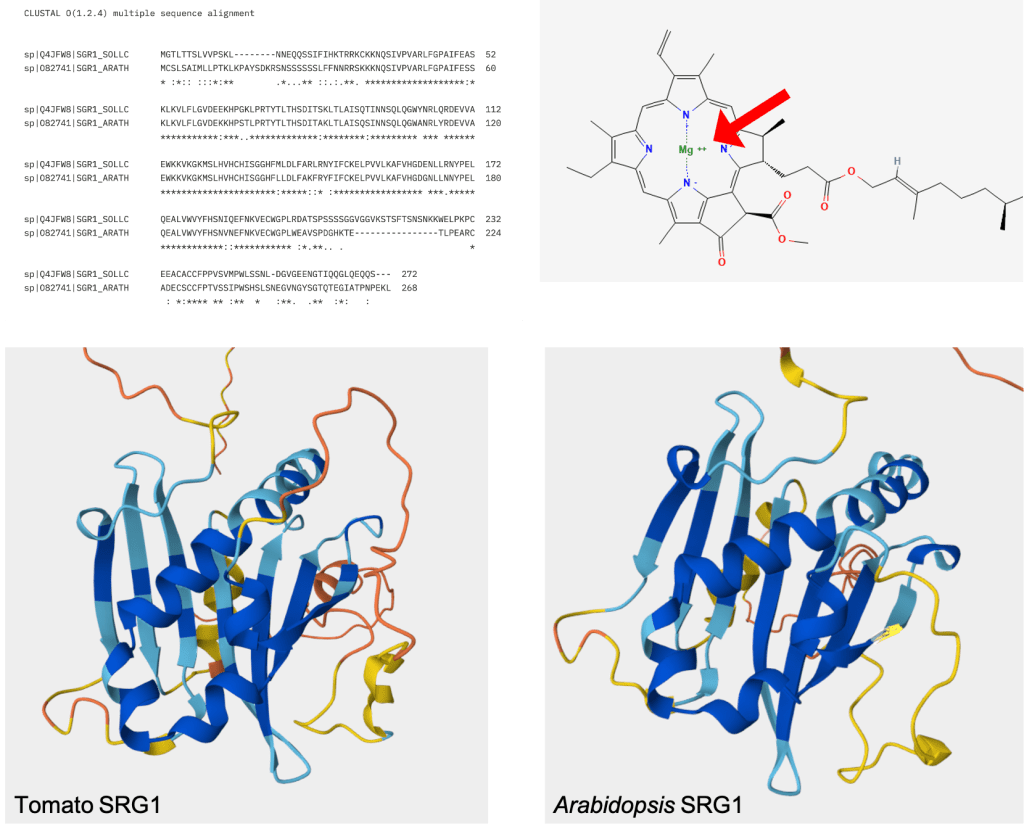
Figure 7. Top Left: Aligned protein sequences of tomato and Arabidopsis SRG1. This alignment was made by inputting the UniProt protein sequences into Clustal Omega (https://www.ebi.ac.uk/jdispatcher/msa/clustalo?stype=protein) with default settings. The two sequences have an extremely high level of similarity, even for orthologs. Top Right: Structure of chlorophyll a from PubChem (https://pubchem.ncbi.nlm.nih.gov/compound/12085802#section=2D-Structure), showing the location of the coordinated magnesium atom within the molecule. As chlorophyll is degraded, this magnesium atom is removed by SRG1. Bottom Left: Alphafold predicted structure of the tomato SRG1 protein, taken from its UniProt entry (https://www.uniprot.org/uniprotkb/Q4JFW8/entry#structure). Bottom Right: Alphafold predicted structure of the Arabidopsis SRG1 protein, taken from its UniProt entry (https://www.uniprot.org/uniprotkb/O82741/entry#structure).
Works cited:
[1] Barry, C.S., McQuinn, R.P., Chung, M., Besuden, A., & Giovannoni, J.J. (2008). Amino Acid Substitutions in Homologs of the STAY-GREEN Protein Are Responsible for the green-flesh and chlorophyll retainer Mutations of Tomato and Pepper. Plant Physiology 147(1), 179-187.
[2] Cheung, A.Y., McNellis, T., & Piekos, B. (1993). Maintenance of Chloroplast Components during Chromoplast Differentiation in the Tomato Mutant Green Flesh. Plant Physiology 101(4), 1223-1229.
[3] Dey, D., Dhar, D., Fortunato, H., Obata, D., Tanaka, A., Tanaka, R., Basu, S., & Ito, H. (2021). Insights into the structure and function of the rate-limiting enzyme of chlorophyll degradation through analysis of a bacteria Mg-dechelatase homolog. Computational and Structural Biotechnology Journal 19, 5333-5347.
[4] Kahlau, S., & Bock, R. (2008). Plastid Transcriptomics and Translatomics of Tomato Fruit Development and Chloroplast-to-Chromoplast Differentiation: Chromoplast Gene Expression Largely Serves the Production of a Single Protein. The Plant Cell 20(4), 856-874.
[5] Rödiger, A., Agne, B., Dobritzsch, D., Helm, S., Müller, F., Pötzsch, N., & Baginsky, S. (2020). Chromoplast differentiation in bell pepper (Capsicum annuum) fruits. The Plant Journal 105(5), 1431-1442.
[6] Roger, A.J., Muñoz-Gómex, S.A., & Kamikawa, R. (2017). The Origin and Diversification of Mitochondria. Current Biology 27(21), PR1177-R1192.
[7] Rosso, S.W. (1968). The ultrastructure of chromoplast development in tomatoes. Journal of Ultrastructure Research 25(3-4), 307-322.
[8] Sadali, N.M., Sowden, R.G., Ling, Q., & Jarvis, R.P. (2019). Differentiation of chromoplasts and other plastids in plants. Plant Cell Reports 38(7), 803-818.
[9] Schweiggert, R.M., Mezger, D., Schimpf, f., Steingass, C.B., & Carle, R. (2012). Influence of chromoplast morphology on carotenoid bioaccessibility of carrot, mango, papaya, and tomato. Food Chemistry 135(4), 2736-2742.
[10] Shimoda, Y., Ito, H., & Tanaka, A. (2016). Arabidopsis STAY-GREEN, Mendel’s Green Cotyledon Gene, Encodes Magnesium-Dechelatase. The Plant Cell 28(9), 2147-2160.
[11] Zimorski, V., Ku, Chuan, Martin, W.F., & Guold, S.B. (2014). Endosymbiotic theory for organelle origins. Current Opinion in Microbiology 22, 38-48.
[12] Zita, W., Bressoud, S., Glauser, G., Kessler, F., & Shanmugabalaji, V. (2022). Chromoplast plastoglobules recruit the carotenoid biosynthetic pathway and contribute to carotenoid accumulation during tomato fruit maturation. PLOS One 17(12), e0277774.